Fluorescence microscopy helps provide new insight into how cancer cells metastasize
By using fluorescence microscopy, scientists at the California NanoSystems Institute at the University of California Los Angeles (UCLA) have discovered an alternate theory on how some cancer cells metastasize—work that could lead to new ways for keeping melanoma from spreading.
Related: Fluorescence microscopy aids in melanoma development discovery
A commonly held theory about how cancer spreads is that tumor cells break off from the primary tumor and travel through the bloodstream to reach other organs, where they attach and grow into new tumors. But questions about that process have remained because circulating tumor cells in the blood sometimes have a short lifespan, and because of a lack of knowledge about how the cells leave the bloodstream and attach to organs.
The research team was led by Laurent Bentolila, director of UCLA's Advanced Light Microscopy/Spectroscopy lab, and included Claire Lugassy and Raymond Barnhill (formerly of UCLA and now of France's Institut Curie). They theorized that—in addition to the prevailing theory about how cancer spreads—tumor cells also could spread through the body by a mechanism called angiotropism, meaning that they could travel along the outside of blood vessels without entering into the bloodstream.
Over the past decade, Lugassy and Barnhill gathered proof that tumor cells, especially those of the deadly skin cancer melanoma, creep along the outside of blood vessels like tiny spiders to spread cancer. They also found that the migrating cancer cells mimicked pericytes—cells that line the capillary blood vessels—which prevented the cancer cells from being killed by the human immune system.
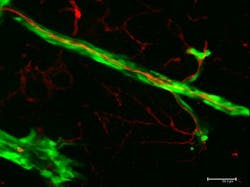
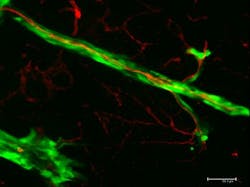
The scientists infused blood vessels with red fluorescent dye while human melanoma cells, which were dyed green, were injected into the brain of a mouse. They used confocal fluorescence microscopy, which provides true 3D optical resolution, to create 3D images in which the dyed tumor cells and the vessels glowed under specific light. The images showed the cells begin to grow as a primary tumor at the injection site. Soon, the researchers observed the green cells spreading from the tumor and migrating along the outer surfaces of the red-dyed blood vessels.
Bentolila explains that if tumor cells can spread by continuous migration along the surfaces of blood vessels and other anatomical structures such as nerves, they now have an escape route outside the bloodstream. If tumor cells are found circulating in the bloodstream, Bentolila says, doctors might prescribe chemotherapy—but if the metastasizing cells are on the outside of the blood vessels, they escape exposure to the treatment and continue to spread cancer.
The findings will enable researchers to seek new targets for deadly cancers such as glioma, glioblastoma, pancreatic cancer, prostate cancer, and gynecological carcinosarcomas.
Full details of the work appear in the journal Scientific Reports; for more information, please visit http://dx.doi.org/10.1038/srep23834.